- English
- 简体中文
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
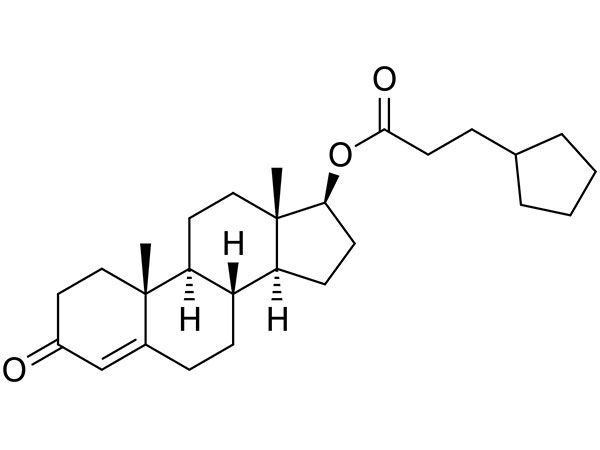
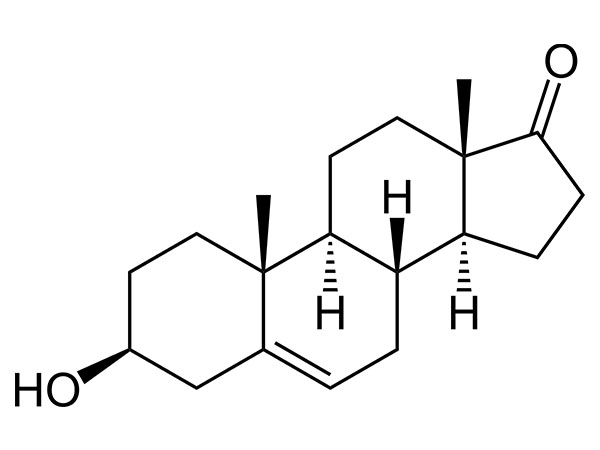
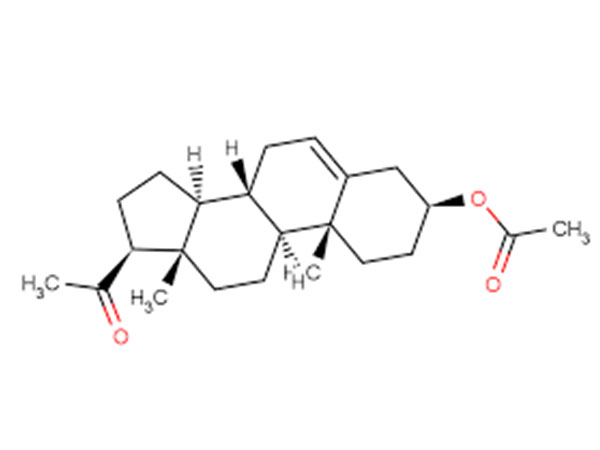
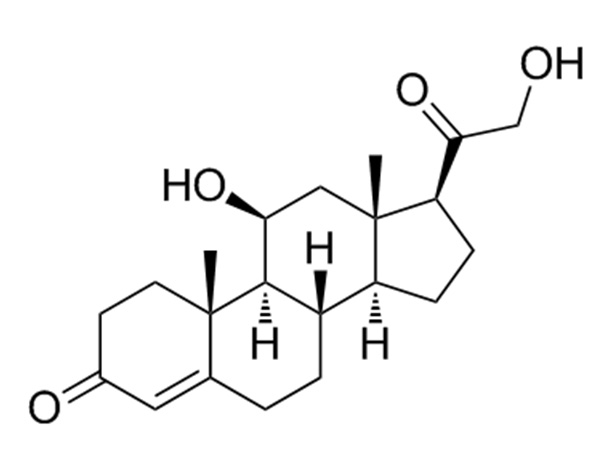
China Prasterone Acetate Manufacturer
Our primary goal is to offer our clients a serious and responsible business relationship, providing personalized attention to all of them for Prasterone Acetate,CAS:853-23-6,DHEA Acetate, Safety by innovation is our promise to each other.
Prasterone Acetate, Our expert engineering team will generally be prepared to serve you for consultation and feedback. We're able to also give you with free of charge samples to meet your requirements. Best efforts will likely be produced to provide you the best service and merchandise. When you are keen on our business and items, make sure you speak to us by sending us emails or call us quickly. In an effort to know our merchandise and company extra, you may come to our factory to view it. We'll generally welcome guests from all over the world to our business to create business relations with us. Be sure to feel cost-free to speak to us for small business and we believe we are going to share the best trading experience with all our merchants.
Hot Products
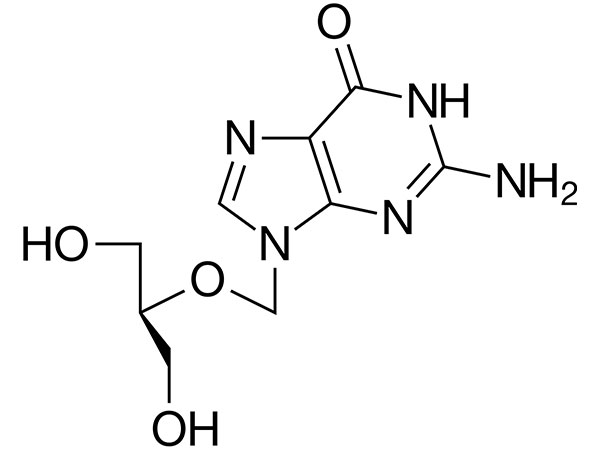
Ganciclovir
Ganciclovir has CP ,EP, USP specification. DMF and GMP approved.CAS:82410-32-0
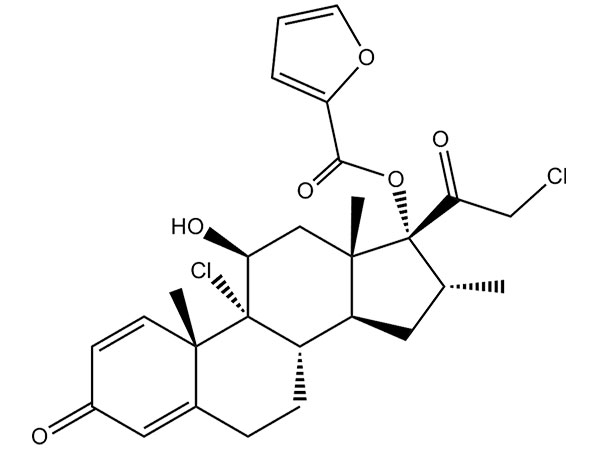
Mometasone Furoate
Mometasone furoate has CP、EP、USP specifications. DMF under filing.CAS:83919-23-7
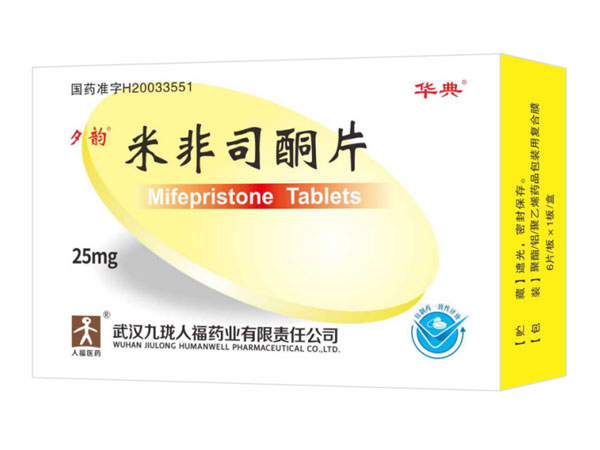
Compound Mifepristone Tablets
Compound Mifepristone Tablets Specifications:Mifepristone 30mg Anorethidrane 5mg*2
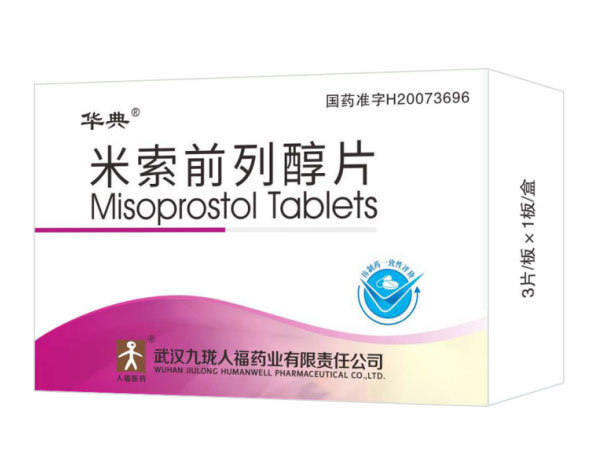
Indications:AbortionMisoprostol Tablets 0.2mg*3
Misoprostol Tablets 0.2mg*3
Specifications:0.2mg*3
Indications:Abortion3-Oxo-4-androsten-17β-carboxylic acid
3-Oxo-4-androsten-17β-carboxylic acid is an intermediate of Dutasteride.CAS:302-97-6
4-aza-5α-androstan-1-ene-3-oxo-17β-carboxylic acid
4-aza-5α-androstan-1-ene-3-oxo-17β-carboxylic acid is an intermediate of Dutasteride.CAS:103335-55-3