- English
- 简体中文
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
China CAS:439239-92-6 Manufacturer
We insist over the principle of enhancement of 'High high quality, Efficiency, Sincerity and Down-to-earth working approach' to offer you with superb assistance of processing for CAS:439239-92-6, Top quality, timely company and Aggressive cost, all win us a superior fame in xxx field despite the international intense competition.
CAS:439239-92-6, Our company has a skillful sales team, strong economic foundation, great technical force, advanced equipment, complete testing means, and excellent after-sales services. Our goods have beautiful appearance, fine workmanship and superior quality and win the unanimous approvals of the customers all over the world.
Hot Products
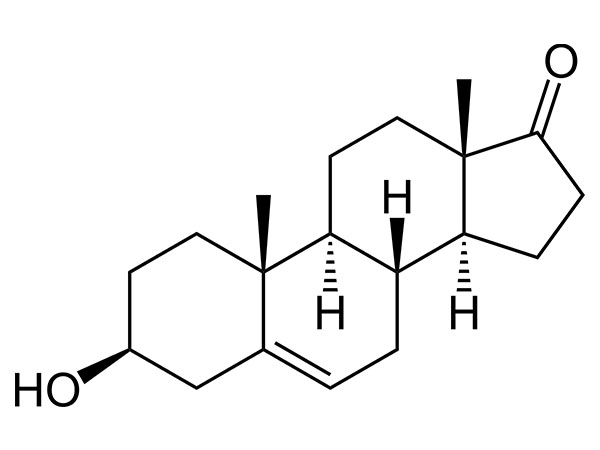
DHEA (Prasterone)
DHEA (Prasterone) has FP and In-house specification. DMF and WC available.CAS:53-43-0
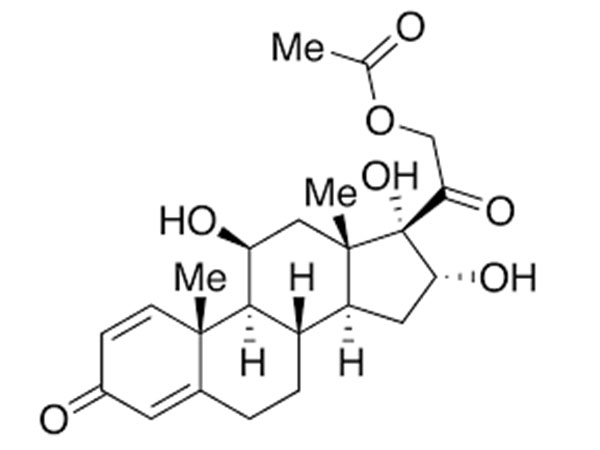
16alpha-Hydroxyprednisolone
16alpha-Hydroxyprednisolone has DMF available.CAS:13951-70-7
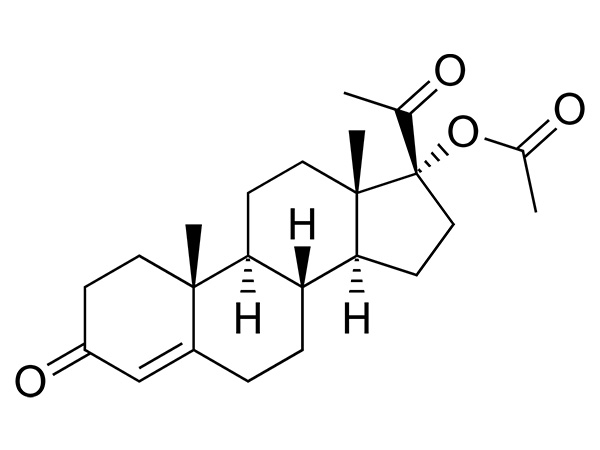
17a-Hydroxyprogesterone Acetate
17a-Hydroxyprogesterone Acetate is an endogenous progesteroid hormone similar to progesterone.CAS:302-23-8
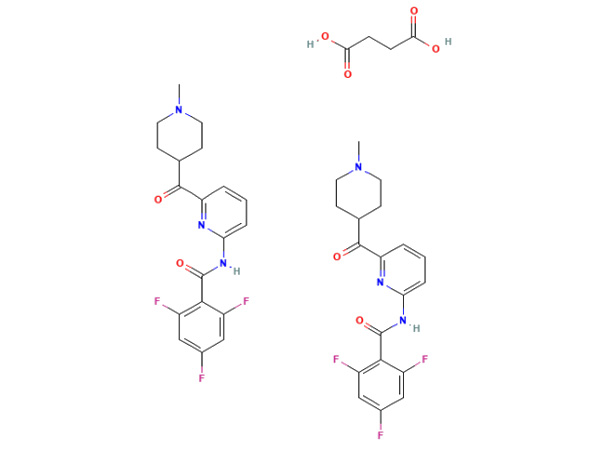
Lasmiditan Succinate
Lasmiditan Succinate has In-house specification. DMF approved.CAS:439239-92-6
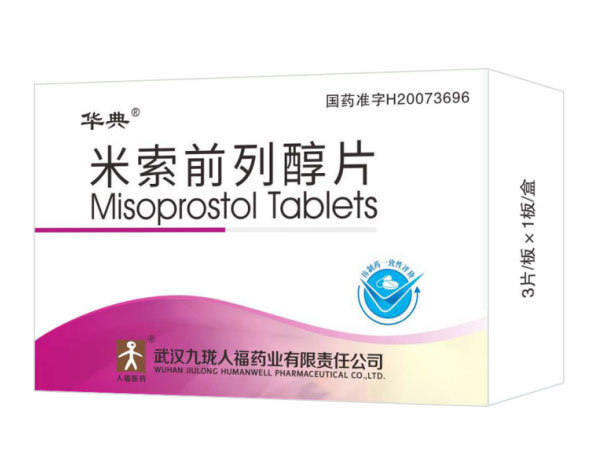
Misoprostol Tablets 0.2mg*3
Misoprostol Tablets 0.2mg*3
Specifications:0.2mg*3
Indications:AbortionAbiraterone
Abiraterone is a steroidal cytochrome P 450 17α-hydroxylase-17,20-lyase inhibitor (CYP17), It is used in combination with prednisone to treat patients with metastatic castration-resistant prostate cancer (prostate cancer that is resistant to medical or surgical treatments that lower testosterone and has already spread to other parts of the body) and metastatic high-risk castration-sensitive prostate cancer.CAS:154229-19-3