- English
- 简体中文
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
China Pregnenolone Manufacturer
Our pursuit and corporation intention is to "Always satisfy our client requirements". We continue to develop and style remarkable high-quality items for each our outdated and new shoppers and accomplish a win-win prospect for our customers likewise as us for Pregnenolone,CAS:1778-02-5, Welcome to setup long-term romantic relationship with us. Best Value Permanently Top quality in China.
Pregnenolone, Insisting over the high-quality generation line management and prospects guide provider, we have made our resolution to offer our shoppers using the initially stage purchasing and soon after provider working experience. Preserving the prevailing helpful relations with our prospects, we even now innovate our product lists the many time to meet up with the brand new wants and stick to the latest trend of this business in Ahmedabad. We're ready to facial area the difficulties and make the transform to grasp many of the possibilities in international trade.
Hot Products
Methylprednisolone
Methylprednisolone has USP, EP, IP, JP and KP specifications. DMF and WC available.CAS:83-43-2
Drospirenone
Drospirenone has EP,USP specifications. DMF approved.CAS:67392-87-4
Ethinyl Estradiol
Ethinyl Estradiol has USP, EP and CP specifications. DMF and GMP available.CAS:57-63-6
Estriol
Estriol has CP, EP, USP specifications. DMF under filing.CAS:50-27-1
Cariprazine hydrochloride
Cariprazine hydrochloride has In-house specification. DMF approved.CAS:1083076-69-0
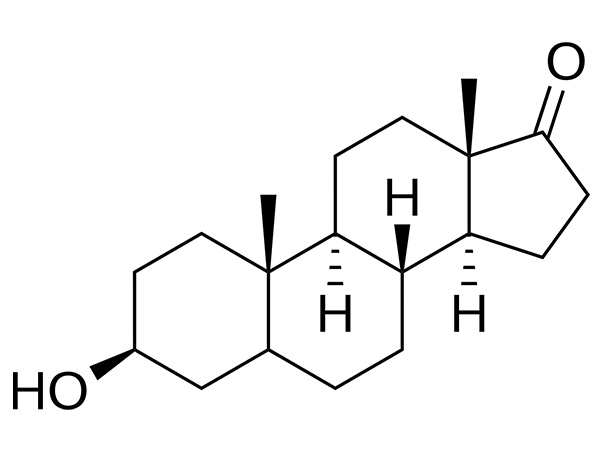
4-aza-5α-androstan-3-oxo-17β-carboxylic acid
4-aza-5α-androstan-3-oxo-17β-carboxylic acid is an intermediate of Dutasteride.CAS:104239-97-6