- English
- 简体中文
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
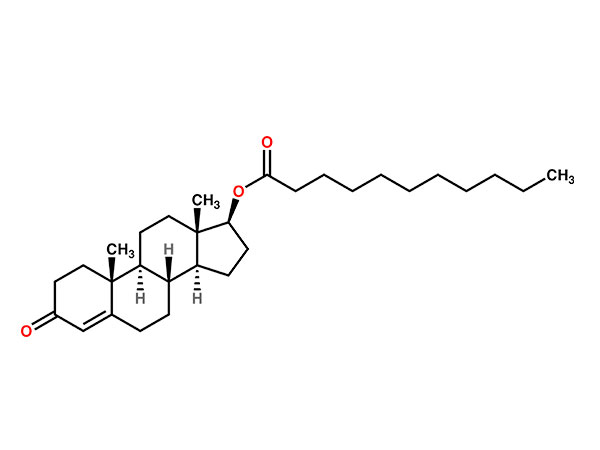
China CAS:58-22-0 Manufacturer
To be a result of ours specialty and repair consciousness, our corporation has won a good popularity amid consumers everywhere in the environment for CAS:58-22-0, We warmly welcome prospects, organization associations and mates from everywhere in the earth to get in touch with us and request cooperation for mutual benefits.
CAS:58-22-0, With high quality, reasonable price, on-time delivery and customized & customized services to help customers achieve their goals successfully, our company has got praise in both domestic and foreign markets. Buyers are welcome to contact us.
Hot Products
Desonide
Desonide has USP and EP specifications. DMF available.CAS:638-94-8
Cyproterone Acetate
For Cyproterone Acetate (CPA), we have CP and EP specifications, CEP/TGA/EU-GMP available.CAS:427-51-0
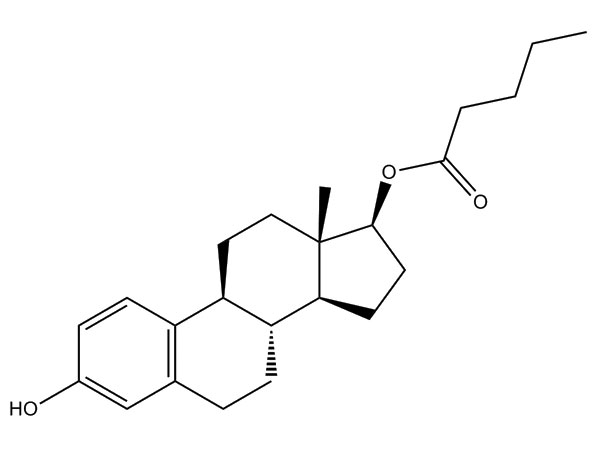
Estradiol Valerate
Estradiol Valerate has CP specification. DMF approved.CAS:979-32-8
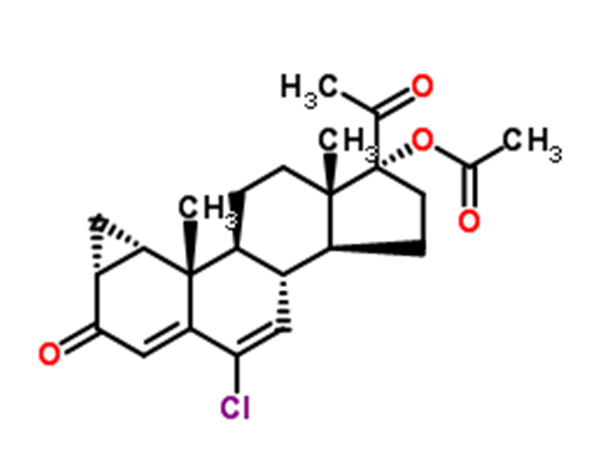
6-chloro-1α-chloromethyl-3,20-dioxo-pregna-4,6-dien-17α-acetoxy
6-chloro-1α-chloromethyl-3,20-dioxo-pregna-4,6-dien-17α-acetoxy is the intermediate of Cyproterone acetate.CAS:17183-98-1
17a-Hydroxyprogesterone Acetate
17a-Hydroxyprogesterone Acetate is an endogenous progesteroid hormone similar to progesterone.CAS:302-23-8
Levonorgestrel Capsules
Levonorgestrel Capsules Specifications:0.75mg*2
Indications:Emergency Contraceptive