- English
- 简体中文
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
China CAS:1778-02-5 Manufacturer
Sticking to the principle of "Super Good quality, Satisfactory service" ,We are striving to become an excellent organization partner of you for CAS:1778-02-5,Pregnenolone, Our goal is always to enable clients comprehend their plans. We have been creating good endeavours to accomplish this win-win scenario and sincerely welcome you to join us.
CAS:1778-02-5, We severely promise that we give all the customers with the best quality products and solutions, the most competitive prices and the most prompt delivery. We hope to win a resplendent future for customers and ourselves.
Hot Products
Ganciclovir
Ganciclovir has CP ,EP, USP specification. DMF and GMP approved.CAS:82410-32-0
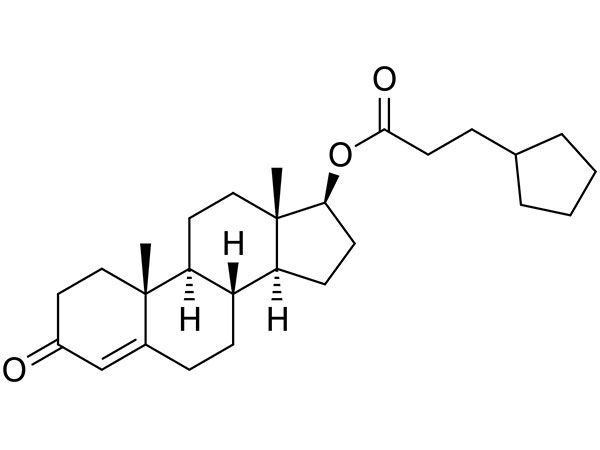
Testosterone Cypionate
Testosterone Cypionate has USP specification. TP available and DMF under filing.CAS:58-20-8
Mono-acetyl Ganciclovir
Mono-acetyl Ganciclovir Specification: Min 97.0%CAS:88110-89-8
Oxcarbazepine
Oxcarbazepine has CP, EP and USP specifications. CEP and DMF available, GMP approved.CAS:28721-07-5
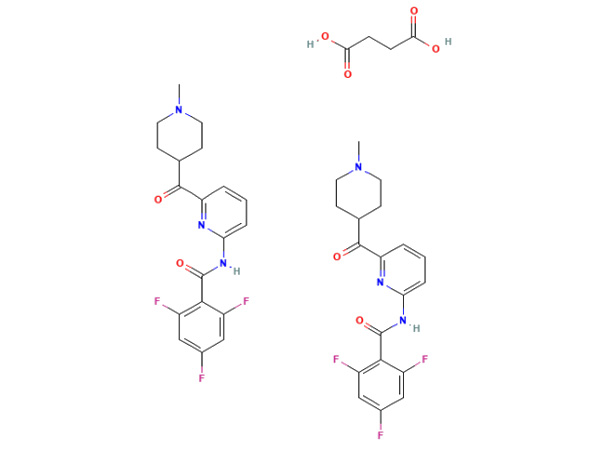
Lasmiditan Succinate
Lasmiditan Succinate has In-house specification. DMF approved.CAS:439239-92-6
Levonorgestrel Capsules
Levonorgestrel Capsules Specifications:0.75mg*2
Indications:Emergency Contraceptive