- English
- 简体中文
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
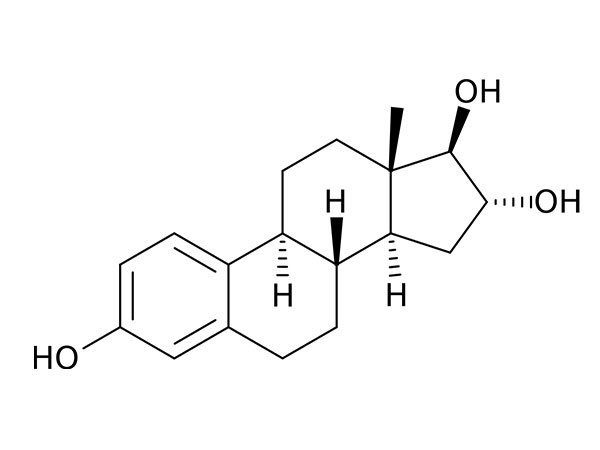
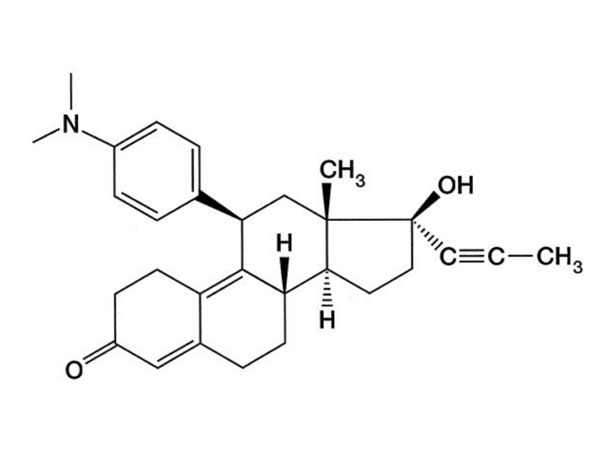
China CAS:107724-20-9 Manufacturer
We've been experienced manufacturer. Wining the majority on the crucial certifications of its market for CAS:107724-20-9, Top quality and competitive rates make our products and solutions appreciate a higher name all around the word.
CAS:107724-20-9, Our products are very popular in the word, like South American, Africa, Asia and so on. Companies to "create first-class products" as the goal, and strive to provide customers with high quality products, provide high-quality after-sales service and technical support, and customer mutual benefit, create a better career and future!
Hot Products
Ethinyl Estradiol
Ethinyl Estradiol has USP, EP and CP specifications. DMF and GMP available.CAS:57-63-6
Estriol
Estriol has CP, EP, USP specifications. DMF under filing.CAS:50-27-1
Cyproterone Acetate Tablets
Cyproterone Acetate Tablets Specifications:50mg*24
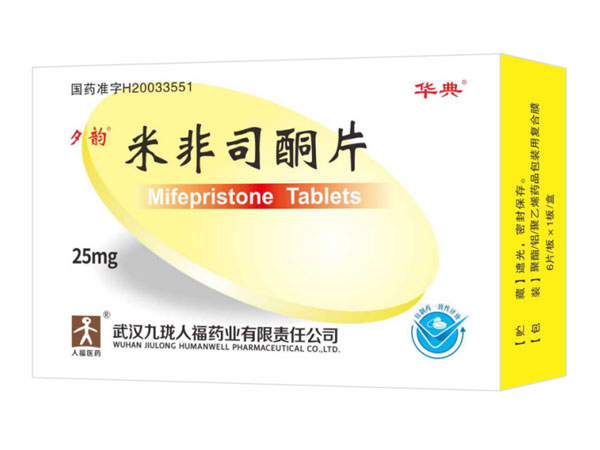
Indications:Anti-androgenMifepristone Tablets
Mifepristone Tablets Specifications:10mg*1
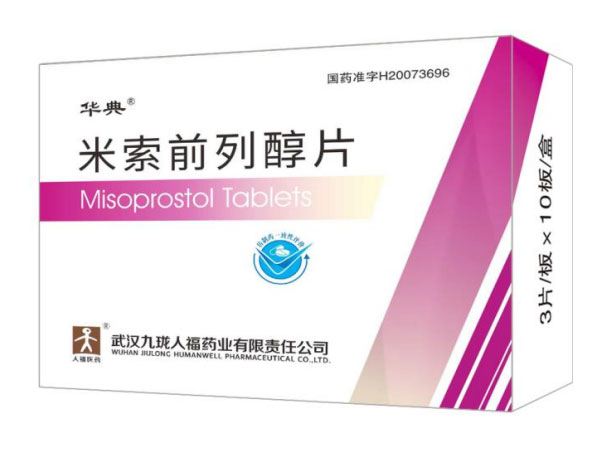
Indications:Emergency ContraceptiveMisoprostol Tablets 0.2mg*30
Misoprostol Tablets 0.2mg*30
Indications:Gastric ulcerEstrone
Estrone is a female sex hormone. The weakest type of estrogen, it’s typically higher after menopause. Like all estrogen, estrone supports female sexual development and function. Low or high estrone can cause symptoms such as irregular bleeding, fatigue or mood swings.CAS:53-16-7