China APIs Category Manufacturer
Gedian Humanwell founded in 2000, with 900 employees, is located in the Thousand Lakes Province - Hubei. The company has two APIs manufacturing sites, one formulation factory and one excipients factory. Gedian Humanwell integrates the reproductive health industry chain from raw materials, active pharmaceutical ingredients (APIs) to finished dosage form(FDF). Our diverse portfolio of products includes Steroidal, Central Nervous System (CNS) and antiviral drugs, etc. Gedian Humanwell is the leading manufacturer and supplier of progesterone, finasteride, oxcarbazepine worldwide, and we are determined to become the market leader in fertility regulation drugs in China, the top supplier of steroidal and CNS APIs in the world.
Gedian Humanwell has a professional, high-level, international R&D and quality management team. For multiple competitive APIs, the company has compiled DMFs and registered in different markets. All our facilities have been inspected and approved cGMP by regulatory authorities including NMPA, USFDA, EDQM, TGA and PMDA etc. Gedian Humanwell provides customers with a high standard of quality management guarantee system.
Gedian Humanwell has a professional, high-level, international R&D and quality management team. For multiple competitive APIs, the company has compiled DMFs and registered in different markets. All our facilities have been inspected and approved cGMP by regulatory authorities including NMPA, USFDA, EDQM, TGA and PMDA etc. Gedian Humanwell provides customers with a high standard of quality management guarantee system.
The domestic sales team of Gedian Humanwell covers 30 provinces in China, while the export business has covered many countries and regions in Europe, North America, South America, Southeast Asia, Middle East and Africa as well as Australia etc.
Active Pharmaceutical Ingredients
| Progestogen and Estrogen | |||
| Product Name | Specification | Approved | CAS No. |
| Progesterone | CP/EP/USP | CEP/EU-GMP/FDA | 57-83-0 |
| Pregnenolone |
In-house |
Kosher | 145-13-1 |
| Medroxyprogesterone Acetate | CP/EP/USP | DMF/GMP | 71-58-9 |
| Megestrol Acetate | CP/EP/USP | DMF in process | 595-33-5 |
| Mifepristone | CP/IP | DMF/GMP | 84371-65-3 |
| Cyproterone Acetate | CP/EP | CEP/TGA/GMP | 427-51-0 |
| Levonorgestrel | CP/EP/USP | DMF/GMP/CEP | 797-63-7 |
| Drospirenone | EP/USP | DMF | 67392-87-4 |
| Gestodene | CP/EP | DMF/GMP | 60282-87-3 |
| Estradiol | CP/EP/USP | DMF | 50-28-2 |
| Estriol | CP/EP/USP | Under R&D | 50-27- 1 |
| Ethinyl Estradiol | CP/EP/USP | DMF/GMP | 57-63-6 |
| Estradiol Valerate | CP | DMF in process | 979-32-8 |
| Tibolone | CP/EP | DMF | 5630-53-5 |
| Dienogest | EP | DMF | 65928-58-7 |
| Fulvestrant | In-house | Under R&D | 129453-61-8 |
| Exemestane | EP/USP | Under R&D | 107868-30-4 |
| Androgen | |||
| Product Name | Specification | Approved | CAS No. |
| Finasteride | CP/EP/USP | CEP/FDA/PMDA/GMP | 98319-26-7 |
| Dutasteride | EP/USP | CEP/DMF | 164656-23-9 |
| Abiraterone Acetate | CP/USP | DMF/WC | 154229-18-2 |
| Testosterone | CP/EP/USP | CEP/DMF/WC | 58-22-0 |
| Testosterone Undecanoate | CP | Under R&D | 5949-44-0 |
| Testosterone Cypionate | USP | Under R&D | 58-20-8 |
| DHEA(Prasterone) | FP/In-house | DMF/WC/Kosher | 53-43-0 |
| Clascoterone | In-house | Under R&D | 19608-29-8 |
| Cortical Hormone | |||
| Product Name | Specification | Approved | CAS No. |
| Budesonide | CP/EP/USP | CEP/FDA/GMP | 51333-22-3 |
| Desonide | USP | DMF | 638-94-8 |
| Eplerenone | EP | CEP/FDA | 107724-20-9 |
| Methylprednisolone | CP/EP/USP | DMF/WC | 83-43-2 |
| Methylprednisolone Hemisuccinate | CP/EP/USP | DMF/WC | 2921-57-5 |
| Fluticasone Propionate | CP/EP/USP | DMF | 80474- 14-2 |
| Fluticasone Furoate | In-house | Under R&D | 397864-44-7 |
| Others | |||
| Product Name | Specification | Approved | CAS No. |
| Oxcarbazepine | CP/EP/USP | CEP/USDMF | 28721-07-5 |
| Eslicarbazepine Acetate | In-house | DMF in process | 236395- 14-5 |
| Ganciclovir | CP/EP/USP | DMF/GMP | 82410-32-0 |
| Valganciclovir Hydrochloride | USP | DMF in process | 175865-59-5 |
| Oseltamivir Phosphate | CP/EP/USP | DMF | 204255- 11-8 |
| Baloxavir Marboxil | In-house | Under R&D | 1985606-14-1 |
| Nintedanib Esylate | In-house | DMF | 656247-18-6 |
| Revefenacin | In-house | DMF in process | 864750-70-9 |
| Mirabegron | In-house | DMF | 223673-61-8 |
| Escitalopram Oxalate | EP/USP/CP | DMF in process | 219861-08-2 |
| Cariprazine Hydrochloride | In-house | DMF in process | 1083076-69-0 |
| Lasmiditan Succinate | In-house | DMF in process | 439239-92-6 |
| Luliconazole | In-house | DMF | 187164-19-8 |
| Apalutamide | In-house | Under R&D | 956104-40-8 |
| Tadalafil | EP/USP/CP | Under R&D | 171596-29-5 |
| Relugolix | In-house | Under R&D | 737789-87-6 |
| Phloroglucinol | CP | DMF | 108-73-6 |
| Ursodeoxycholic Acid | EP/USP/JP | Under R&D | 128-13-2 |
- View as
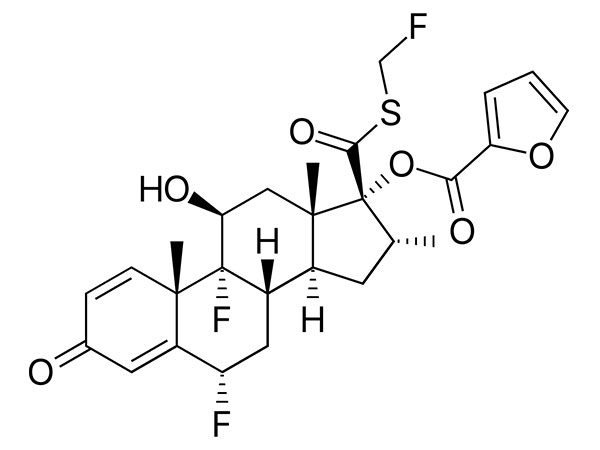
Fluticasone Furoate
Fluticasone furoate has In-house specifications. DMF under filing.
CAS:397864-44-7
Read MoreSend InquiryMometasone Furoate
Mometasone furoate has CP、EP、USP specifications. DMF under filing.
CAS:83919-23-7
Read MoreSend InquiryEslicarbazepine Acetate
Eslicarbazepine Acetate has In-house specification. DMF under filing.
CAS:236395-14-5
Read MoreSend InquiryValganciclovir Hydrochloride
Valganciclovir Hydrochloride has USP specification. DMF under filing.
CAS:175865-59-5
Read MoreSend InquiryOseltamivir Phosphate
Oseltamivir Phosphate has CP ,EP and USP specifications. DMF available.
CAS:204255-11-8
Read MoreSend InquiryRevefenacin
Revefenacin has In-house specification. DMF under filing.
CAS:864750-70-9
Read MoreSend Inquiry
Humanwell Pharmaceutical is one of the largest API manufacture in China. With more than 20 years of experience, we develop, manufacture and trade steroid APIs, intermediates and formulations. Our market covers all over the world, we have strong presences in North America, Europe, South America and Africa, with products sold to more than 150 countries.